Designing Reactions with Post-Transition-State Bifurcations
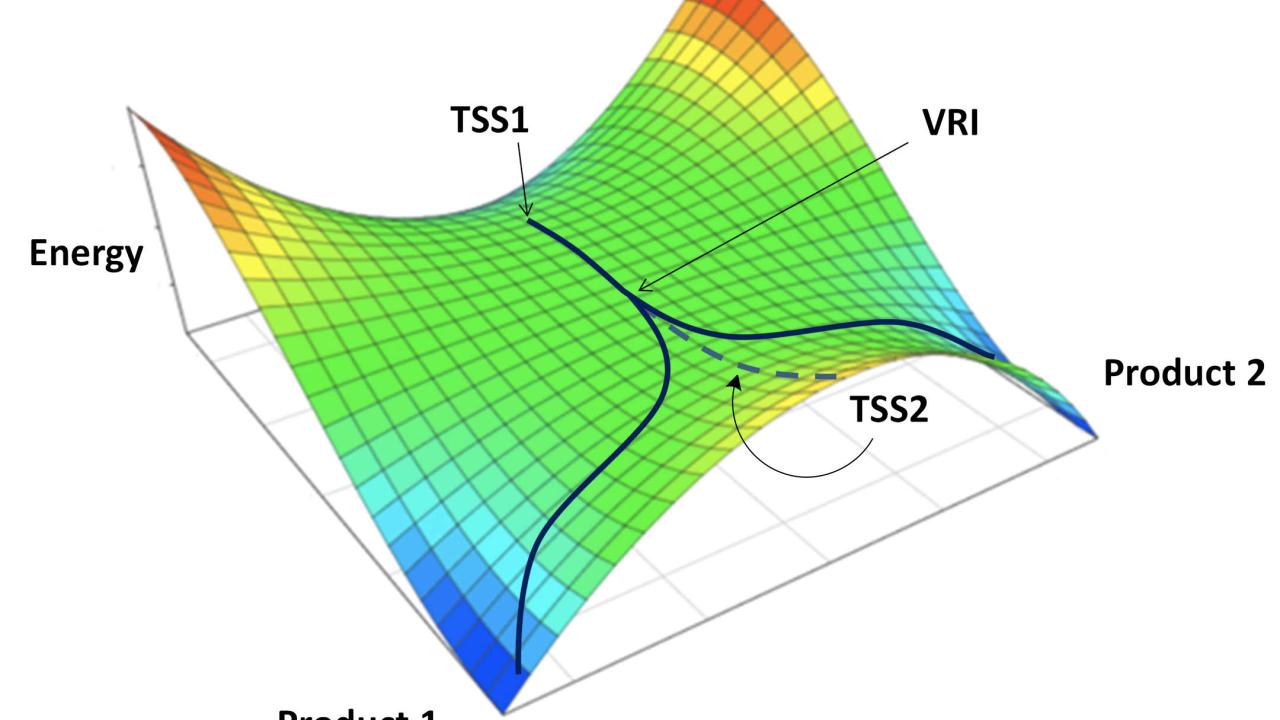
Deep understanding of the factors controlling reactivity and selectivity for chemical reactions empowers chemists to control product distributions and design reactions with desired outcomes. The work described here provides a recipe for designing reactions with post-transition-state bifurcations (PTSBs)—reactions in which a single transition state leads directly to two (or more) products. This type of reaction does not conform to the usual pictures of reactivity presented to students but has recently reared its head in fields as diverse as organometallic chemistry and natural products biosynthesis. By demonstrating that such a reaction can be designed rationally, we exert a degree of dominance over the lack of available data on such reactions, but our ability to design does not guarantee that we fully understand. Although we provide a working model for controlling selectivity for reactions with PTSBs, we hope it will be refined through future work in pursuit of deeper understanding.
This research has also been highlighted recently in American Scientist.
Read the original article here: https://doi.org/10.1016/j.chempr.2018.10.019
